| Unit 8 | Haloalkanes class 12 |
| 8.1 | Nomenclature |
| 8.2 |
Classification |
| 8.3 | Isomerism |
| 8.4 | Preparation (alkanes, alkenes, and alcohols) |
| 8.5 | Physical properties |
| 8.6 | Chemical properties (SN1 and SN2) |
| 8.7 | Formation (alcohol, nitrile, amine, ether, thioether, carbylamine, nitrite, and nitroalkane) |
| 8.8 | Elimination Reaction ( Saytzeff ’s rule) |
| 8.9 | Reaction with Sodium metal (Wurtz's Reaction) |
| 8.10 | Reduction Reaction of Haloalkane |
| 8.11 | Preparation of Chloroform |
| 8.12 | Physical properties of Chloroform |
| 8.13 | Chemical properties of Chloroform |
%20Class%2012%20notes%20NEB%20Nepal.jpg)
|
| Class 12 notes: Haloalkanes with important Questions |
Table of Contents
Unit 8: Haloalkane class 12
IntroductionHaloalkanes are the derivatives of hydrocarbon because they are derived by replacing hydrogen with halogen atoms, therefore the halogen derivatives of alkane are called haloalkanes. Or
The organic compound containing halogen atom (X=-F,-Cl, -Br, -I) as a functional group is called Haloalkanes.
They are also called Alkyl halides.
Uses: Solvent, Medicine, Insecticide, etc.
Haloalkanes are formed by the replacement of one or more hydrogen atoms of alkane by the same number of halogen atoms and are bonded with the carbon atom of alkane through a strong covalent bond. They are presented by the general molecular formula
CnH2n+1X.
Nomenclature of haloalkanes
Prefix: side chain branches substituents
Word root: number of C-atoms
Primary suffix: nature of C-atoms (-ane or -one or -one)
| Formula | Common name (Alkyl+halide) | IUPAC name (Halo +word) |
|---|---|---|
| R-X | alkyl halide | halo alkane |
| CH₃F | methyl fluoride | fluoro methane |
| CH₃CH₂-Cl | ethyl chloride |
chloro ethane |
| CH₃CH₂CH₂Br | propyl bromide | 1-Bromo propane |
| CH₃CH₂-I | ethyl iodide | iodo ethane |
| CHCl₃ | chloroform |
tri chloro methane |
| CHI₃ | iodoform |
tri iodo methane |
| CCl₄ | carbon tetrachloride | tetra chloro methane |
| CH₃-CH-Cl₂ |
ethylidene chloride (geminal dichloride) |
1,1-dichloro ethane |
| Cl-CH₂-CH₂-Cl |
ethylene dichloride (Vicinal dichloride) |
1,2-dichloro ethane |
| CH₃CH₂CH₂Cl | n-propyl chloride | 1-Chloro propane |
|
|
tertiary butyl bromide (Neo-butyl bromide) |
2-Bromo, 2-methyl propane |
| CH₃CH₂CH₂CH₂Br | n-butyl bromide | 1-Bromo butane |
| iso-butyl bromide | 1-Bromo-2-methyl propane | |
| isopropyl chloride | 2-chloro propane | |
| secondary butyl bromide | 2-Bromo butane |
Classification of haloalkanes
[A] Based on the nature of the carbon atom
1. Primary haloalkane (1 ̊)
The haloalkane in which the halogen-containing carbon is further bonded to one carbon atom (one alkyl group) or primary carbon is called primary haloalkane.
|
|
|
Primary haloalkane |
2. Secondary haloalkane (2 ̊)
The haloalkane in which the halogen-containing carbon is further bonded to two carbon atoms (two alkyl groups) or secondary carbon is called secondary haloalkane.
|
|
|
Secondary Haloalkane |
3. Tertiary haloalkane (3 ̊)
The haloalkane in which the halogen-containing carbon is further bonded to three carbon atoms (three alkyl groups) or tertiary carbon is called tertiary haloalkane.
|
|
|
Tertiary Haloalkane |
[B] based on the number of halogen atoms
1. Mono-halo alkane
Haloalkane contains only one halogen atom.
|
|
|
Mono-haloalkane |
2. Di-haloalkane
Haloalkane contains two halogen atoms.
|
|
|
Di-haloalkane |
3. Poly-haloalkane
Haloalkane contains three or more halogen atoms.
|
|
|
Poly-haloalkane |
Isomerism in haloalakanes
1. Chain isomerism
Haloalkanes have the same number of carbon atoms but the different number in carbon chain length is called chain isomers.
|
|
|
chain isomerism: Bromo pentane |
2. Position isomerism
Haloalkanes have the same molecular formula but the different positions of halogen atoms on the carbon chain are called position isomers.|
|
|
position isomerism: chloropropane |
Self-test:
Q.Write all possible isomers with the molecular formula C4H9I and give their IUPAC name.
General methods of preparation of haloalkane
1. From alkanes (Halogenation of alkanes)
The haloalkanes are prepared by treating alkane with a limited amount of halogen in presence of halogen carriers and sunlight or heat.
|
|
|
Formation of haloalkane from an alkane |
On excess supply of chlorine poly-substituted product is formed.
|
|
|
methane to tetra chloromethane in excess of chlorine |
In the case of higher alkanes two or more possible products are formed.
|
|
|
higher alkanes give more than one products |
The bromination is carried out in presence of FeBr₃ under sunlight or heat.
The iodination of an alkane is a reversible reaction. So to obtain iodoalkane strong oxidizing agent like conc.HNO₃ or HIO₃ is used to increase the rate of the forward reaction.

|
|
A reaction mechanism for iodoalkane from an alkane |
Thus, formed iodine increases the rate of forwarding reaction. Hence the iodination of alkane must be carried out in presence of a strong oxidizing agent.

|
|
The reaction of formation of iodomethane from methane in presence
of conc.HNO3 |
2. From alkenes(Hydrohalogenation of alkenes)
The haloalkanes are prepared by the reaction of an alkene with halogen acid (HF, HCl, HBr, HI). The reaction is called the Hydrohalogenation reaction.
If an unsymmetrical alkene is taken then two possible
products are formed.
The Formation and stability of these two products can be explained by following two rules:
[A] Markovnikov’s rule:
According to this rule “when an unsymmetrical alkene reacts with an unsymmetrical reagent than the positive part of reagent goes to that double bonded carbon containing greater number of the hydrogen atoms”. For example
[B] Peroxide effect (Anti- Markovnikov’s rule)
According to this rule “when an unsymmetrical alkene reacts with the unsymmetrical reagent in presence of
organic peroxide
(R-O-O-R)then the positive part of the reagent goes to that double bonded carbon containing less number of
the hydrogen atom”. For example,

|
|
Anti-Markovnikov's reaction |
It is also called the Kharasch effect.
HCl and HI do not give Markovnikov’s addition, why?
Ans: H-Cl is highly polar and hence does not undergo hemolysis easily. HI undergoes homolysis to give iodine free radicals which instantly combine to give I2.
3. From Alcohol
Generally, haloalkanes are prepared by the reaction of alcohols with haloacids or phosphorous halide, or thionyl chloride.
(a) Reaction with halogen acid(HX)
The chloroalkane is prepared by the reaction of alcohol with HCl in presence of anhydrous zinc chloride (ZnCl₂).
The mixture of conc. HCl and anhydrous ZnCl₂ is called Lucas reagent.
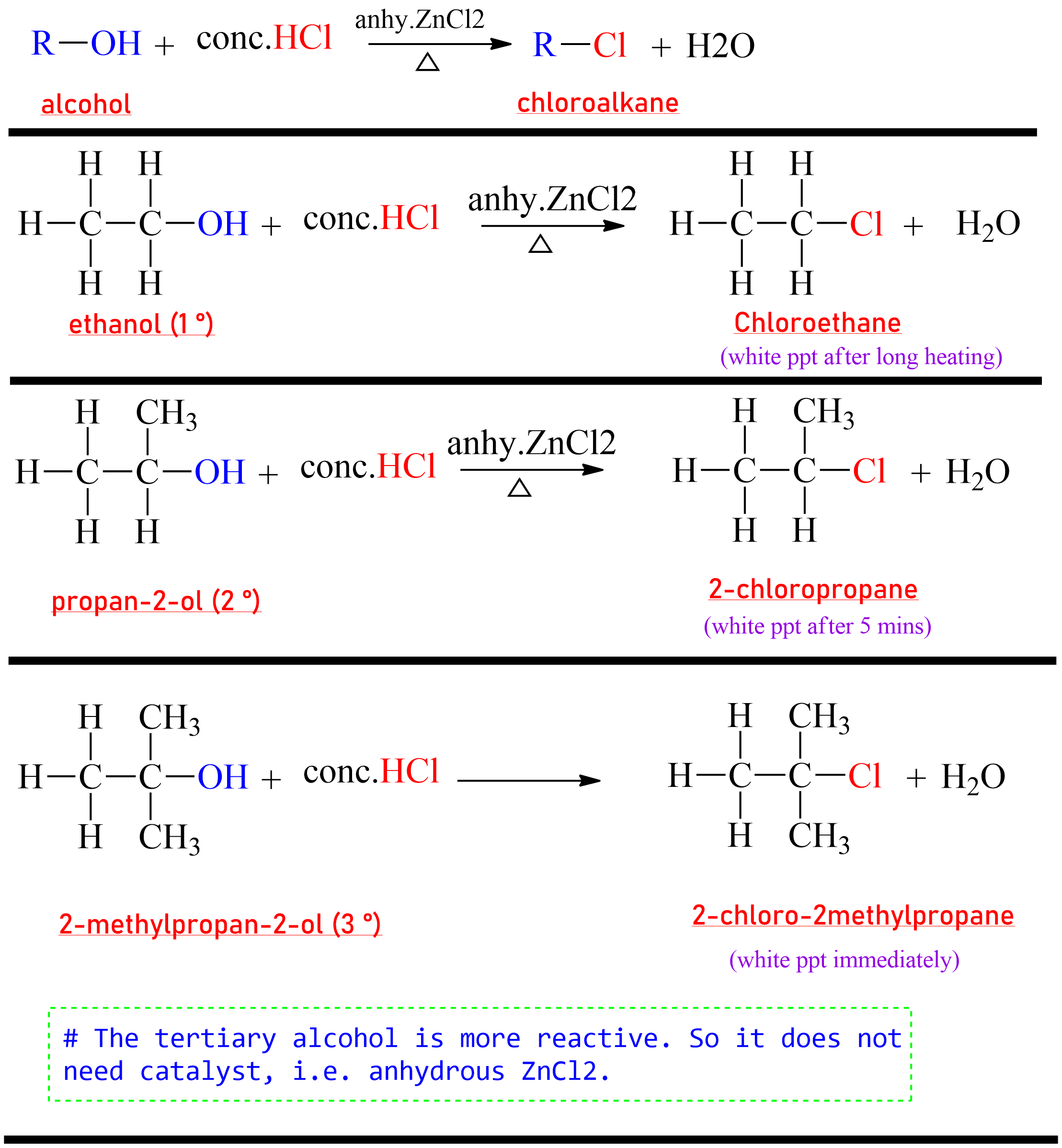
|
|
A reaction from 1,2,3 degree alcohol to respective Chloro
substituted product. |
(b) Reaction with a phosphorous halide (PX₅ or PX₃)
The haloalkanes are prepared by the action of alcohol with PX₅ or PX₃.
Since PBr₃ and PI₃ are unstable compounds. So they are prepared in the reaction mixture (In-situ form) by the action of red phosphorous with Br₂ or I₂.
(c) Reaction with thionyl chloride (SOCl₂)
(Darzen’s reaction)
The chloroalkanes are prepared by heating alcohol with SOCl₂ in presence of pyridine. Only chloroalkane is prepared by this method. From this method, pure chloroalkane can be prepared because SO₂ and HCl evolved as gases.

|
|
Darzen's Reaction or Reaction of alcohol with thionyl
chloride. |
Physical properties of haloalkanes
- Lower members of haloalkane methyl chloride and methyl bromide are colorless gases, higher are colorless and sweet-smelling liquids and next higher are colorless solids.
- They are insoluble in water and soluble in almost all organic solvents like ether, alcohol, etc.
- They burn with green-edged flame in the air.
- The boiling point of haloalkanes is higher than corresponding parent alkanes.
- The boiling point of haloalkane having the same alkyl group is RI>RBr > RCl due to the large size of the halogen atom.
- Branched-chain haloalkane has a lower boiling point than straight-chain haloalkane due to its spherical nature.
- The B.P. increase as the increase in the alkyl group.
Chemical properties of haloalkanes
The haloalkanes are more reactive than alkanes due to the presence of polar C-X bonds. The polarity arises due to the difference in electronegativity value between carbon and a halogen atom.
[A] Nucleophilic substitution reaction
The nucleophile is electron-rich species having lone pairs of electrons or negative charges and can attack to electron-deficient center. When a nucleophile is substituted by another nucleophile then the reaction is called a nucleophilic substitution reaction.
Nuc: + R-LG → R-Nuc + LG:
Nuc- nucleophile LG- Leaving group
Example: R-Br + OH− → R-OH + Br−
Here, the existing nucleophile has been substituted by an incoming nucleophile.
The alkyl halides undergo nucleophilic substitution reaction (SN- reaction ) by following two mechanisms.
S stands for Substitution
N stand for Nucleophile
The number represents Kinetic order
SN1 reaction:
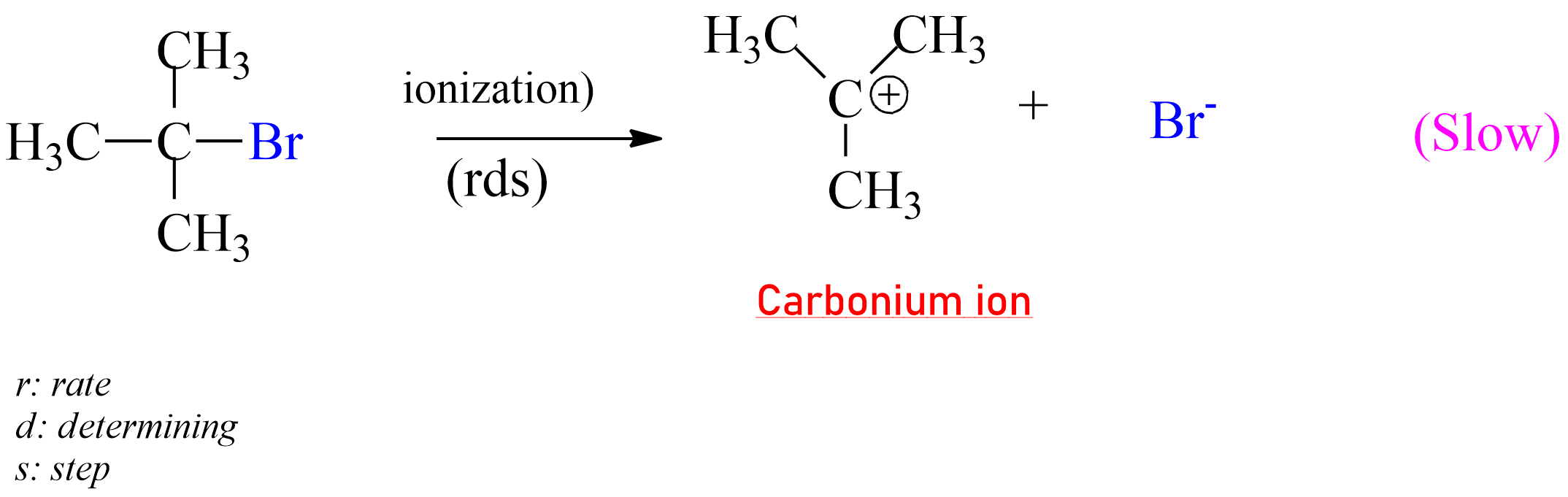
|
|
The rate-determining step of SN1 Reaction |
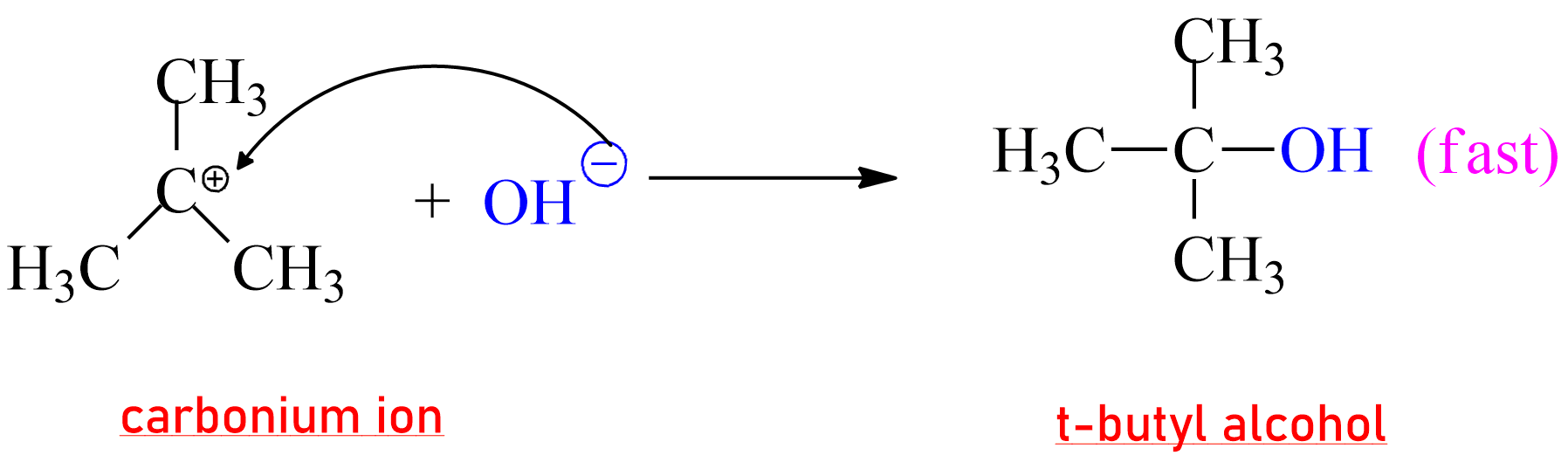
|
|
Formation of product in SN1 Reaction |
SN2 reaction:

Difference Between SN1 and SN2 Reaction
| SN1 | SN2 |
| This follows the Unimolecular Rate of Reaction mechanism | This follows the Bimolecular Rate of Reaction mechanism |
| Follows 1st order Kinetic Reaction | Follows 2nd order Kinetic Reaction |
| Two-Step Mechanism | One Step Mechanism |
| A carbocation is formed as an intermediate part | No Carbocation |
| Racemization occurs | Inversion occurs |
| Order: 3°>2°>1° | Order: 1°>2°>3° |
| RoR∝ [ substituent] | RoR∝ [ substituent].[ Nucleophile] |
1. Reaction with aqueous NaOH or KOH (Formation of alcohol)
When
haloalkane
reacts
with an aqueous
solution
of
NaOH or KOH
then
alcohol
is
formed.
Ambident Nucleophile
Similarly, NO₂⁻has two attacking sites.
R-X+CN⁻ ⟶ R-CN (Attack by carbon)
R-X+CN⁻ ⟶ R-NC (Attack by nitrogen)
2.
Reaction with alcoholic NaCN or
KCN
When haloalkane is heated with an alcoholic solution of NaCN or KCN then alkane nitrile (Cyanides) are formed. This reaction is largely used to increase the number of carbon atoms during organic conversion.
Alkane nitrile(Cyanides) is a beneficial chemical that gives various products when treated with different reagents.
(a) Reduction Reaction
A reaction in which an organic nitrile is reduced by nascent hydrogen (e.g. from sodium in ethanol) to a primary amine is REDUCTION REACTION.
b) Complete hydrolysis
c) partial hydrolysis
Q. Convert methane to ethanoic acid
3.
Reaction
with
alcoholic AgCN
When haloalkane is heated with an alcoholic solution of AgCN then alkyl isocyanide is formed.
Here, AgCN is a covalent compound. So it does not dissociate easily. Therefore the lone pair of electrons in the nitrogen atom attacks haloalkane to form isocyanides.
Similarly, isocyanide forms different compounds as:
(a)Reduction
(b)Acidic hydrolysis
4. Formation of amines
When haloalkane is heated with alc. ammonia then amines are formed.
5. Reaction with sodium alkoxide(R-ONa) (Williamson’s ether synthesis/ Formation of ether)
When haloalkane is heated with sodium alkoxide then ether is formed. This reaction is called Williamson’s etherification reaction.
Both symmetrical and unsymmetrical ether can be prepared by this method.
6. Formation of thioether
Thioether is (R-S-R”) analog of ether.
Name sulfides like ethers, replacing ”sulfide” for “ether” in the
common name, or “alkylthio” for “alkoxy” in the IUPAC system.
7. Carbylanime
When primary animes are heated with alcoholic potassium hydroxide (KOH) and chloroform forms a product which is a foul-smelling substance called carbylanime reaction.
8. Reaction with aqueous NaNO₂ or KNO₂
When haloalkanes are heated with aqueous NaNO₂ or KNO₂ solution then alkyl nitrite is formed.
Here, NaNO₂ is an ionic compound. Hence Na−O bond breaks easily and negatively charged oxygen attacks haloalkane to form alkyl nitrite.[Na-O-N=O]→[Na⁺+ ONO⁻]
9. Reaction with alcoholic silver nitrite (AgNO₂)
When haloalkanes are heated with alcoholic AgNO₂ solution then nitroalkanes are formed.
Here, AgNO₂ is a covalent compound. Hence the lone pair of electrons on the nitrogen atom attacks an alkyl group of haloalkane to form nitroalkane.[Ag-O-N=O]→[Ag−NO₂]
[B] Elimination reaction (ᵦ- elimination reaction) Dehydrohalogenation reaction
When haloalkane is boiled with an alcoholic solution of NaOH or KOH then alkene is formed. In this reaction, one hydrogen and halogen atom are removed from adjacent carbon. So this reaction is called dehydrohalogenation reaction.
This reaction is also called β-elimination or 1,2-elimination.
Saytzeff’s rule
If dehydrohalogenation of haloalkane gives two or more alkenes then alkene containing a greater number of the alkyl group on double bonded carbon is the major product. This rule is called Saytzeff ’s rule.

|
| Saytzeff's rule |
[C] Reaction with metal
Reaction with sodium (Wurtz’s reaction)
When alkyl halide is heated with sodium metal in presence of dry ether, then alkane having a double number of carbon atoms is formed. Therefore it is used to increase carbon length.

|
|
Reaction with sodium ( Wurtz's reaction ) |
[D] Reduction reaction of Haloalkane
1. Catalytic reduction
2. Reduction with metal hydride
3. Reduction with metallic solution
- Sn-Zn-Fe/HCl
- C₂H₅OH/Na
- Red.P₄/HI
Preparation of Chloroform
The chloroform is prepared in the lab by heating ethanol or acetone with the aqueous paste of bleaching powder. In this process, the bleaching powder acts as an oxidizing agent, chlorinating agent as well as the hydrolyzing agent.
1. From ethanol (ethyl alcohol)
(a) Oxidation of ethanol:
Chlorine oxidizes ethanol into ethanal.
(b) Chlorination of ethanal:
Excess chlorine reacts with ethanal to give chloral.
(c) Hydrolysis of chloral:
The chloral is hydrolyzed with Ca(OH)₂ to form chloroform.
2. From propanone (acetone)
(a) Chlorination of acetone
Acetone is chlorinated by passing excess chlorine through forming tri chloroacetone.
(b) Hydrolysis of tri chloroacetone
At last, tri chloroacetone is hydrolyzed with calcium hydroxide to form chloroform.
Physical properties of Chloroform
- It is a colorless mobile oily liquid.
- Its boiling point is 61 ̊C and melting point is -60 ̊C.
- It is heavier than water having an sp. gravity of 1.48.
- It has a characteristic sweet smell and taste.
- It is insoluble in water and soluble in almost all organic
- solvents like benzene, ether, etc.
- It is a good solvent for dissolving fats, oils, resins, waxes, etc.
- The Vapour of chloroform causes temporary unconsciousness when taken in small amounts. So, it is used as an anesthetic drug. *Why it happen?
Chemical properties of chloroform
1. Action with air (Oxidation)
When chloroform is exposed to air in presence of sunlight, a highly poisonous phosgene gas (Carbonyl chloride) is formed.
Therefore, the following precautions are taken while storing the chloroform.
- It is always stored in a dark brown colored bottle to protect it from sunlight.
- It is completely filled up in the bottle to keep out air.
- A small amount (1%) of ethyl alcohol is added to the bottle of chloroform. The ethyl alcohol reacts with phosgene gas formed during storage to give non-toxic harmless diethyl carbonate.

2. Reaction with silver powder
When chloroform is heated with silver powder then acetylene is formed.
3. Reaction with conc.HNO₃(Nitration)
When chloroform is treated with conc. nitric acid then chloropicrin is formed which is used as an insecticide and war gas (Tear gas).
4. Reaction with acetone
When chloroform is the heated presence of aqueous NaOH or KOH then with acetone in a crystalline solid of chloretone is formed which is used hypnotic drug (sleep-inducing).

5. Reduction
i)Acidic medium(Zn/HCl)
When chloroform is reduced in an acidic medium, methylene chloride is formed.
ii)Basic medium(Zn/H₂O)
On reduction in neutral medium, methane is formed.
Purity test of chloroform
6. Reaction with silver nitrate (AgNO₃)
i) With Pure Chloroform
Pure chloroform does not give white ppt. with AgNO₃ solution because the C-Cl bond in chloroform is a strong covalent bond and can not give Cl⁻ ion.
ii) With impure Chloroform
But, impure chloroform gives white ppt. of AgCl with AgNO₃ due to the presence of Cl⁻ ion after the oxidation of chloroform in the air.


Read More: Class 12 Chemistry
Important Question For Haloalkane and Chloroform

|
| Fig: Haloalkanes whole syllabus/course content |
Frequently Asked Questions
What do you mean by haloalkanes?
Haloalkanes and haloarenes are hydrocarbons in which one or more hydrogen atoms have been replaced with halogen atoms.
What haloalkanes called?
The haloalkanes, also known as alkyl halides, are a group of chemical compounds comprised of an alkane with one or more hydrogens replaced by a halogen atom (fluorine, chlorine, bromine, or iodine).
What are haloalkanes with examples?
The haloalkanes are a group of chemical compounds comprised of an alkane with one or more hydrogens replaced by a halogen atom (fluorine, chlorine, bromine, or iodine). Example: Chloroethane (CH3CH2Cl).
What are the uses of haloalkanes and Haloarenes?
Haloalkanes and haloarenes are used for many industrial and day to day purposes. They are used as flame retardants, propellants, solvents, pharmaceuticals, refrigerants, fire extinguishants, and many more. They are used as solvents for non-polar compounds.
How many types of haloalkanes are there?
Halogenoalkanes are also called haloalkanes or alkyl halides. All halogenoalkanes contain a halogen atom - fluorine, chlorine, bromine or iodine - attached to an alkyl group. There are three different kinds of halogenoalkanes: Primary, secondary and tertiary.
Why haloalkanes are more reactive than Haloarenes?
Greater the dipole moment, greater is the polarity and more is the reactivity. Thus, C−X bond of haloalkane is more polar than the C−X bond of haloarene. Hence, haloarenes are less reactive than haloalkanes.
What is the difference between haloalkanes and Haloarenes?
When hydrogen atoms in aliphatic hydrocarbons(alkanes) are replaced by halogen atoms ,the compounds formed are known as haloalkanes. Similarly, when hydrogen atoms attached to benzene rings are replaced by halogen atoms the compounds that are formed are known as haloarenes.
What is the haloalkanes formula?
CnH2n+2=Alkanes, CnH2n−2=Alkynes, CnH2n=Alkene, and CnH2n+1X is haloalkanes.



